Product Specification
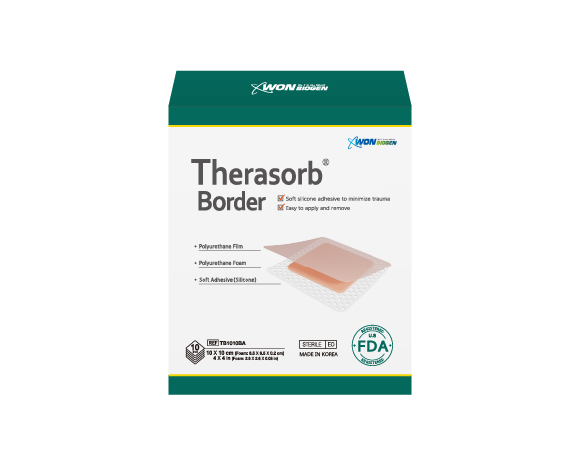
Therasorb Border
- Classfication
- Quasi-Drug
- Size
- 5.5 x 6.5cm (3 x 4 x 0.2cm)
7.5 x 7.5cm (5 x 5 x 0.2cm)
9 x 15cm (5 x 10 x 0.2cm)
9 x 25cm (5 x 20 x 0.2cm)
10 x 10cm (6.5 x 6.5 x 0.2cm)
- Efficacy
- Absorption and protection of secretions from wounds
- Usage
- Wash the affected area (washing with saline, etc.) and attach the wound contact layer in direct contact with the affected area. Change the dressing if the secretions are excessively absorbed.
- Precautions
- If this quasi-drug is used together with a hydrochlorite solution (Eusol, etc.) or an oxidizing agent such as hydrogen peroxide, be careful as the foaming filler may be destroyed.
- Exp. Date
- 36 months from date of manufacture
- Storage
- Store at room temperature (1~30℃), airtight container